

|
Indications |
Diethylstilbestrol is one of the older synthetic estrogens in use. It was used to treat prostate cancer but is now rarely used for this purpose because of its adverse effects,although it is occasionally used in postmenopausal women with breast cancer. It is taken orally in tablet form. |
|
Definition |
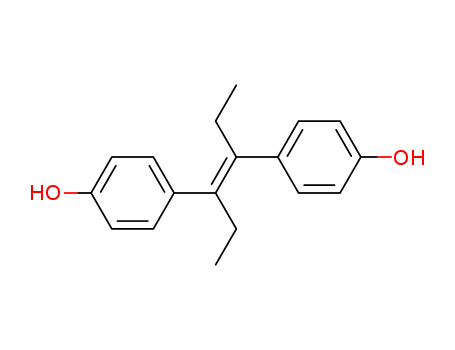
ChEBI: Diethylstilbestrol is an olefinic compound that is trans-hex-3-ene in which the hydrogens at positions 3 and 4 have been replaced by p-hydroxyphenyl groups. It has a role as an antineoplastic agent, a carcinogenic agent, a xenoestrogen, an EC 3.6.3.10 (H(+)/K(+)-exchanging ATPase) inhibitor, an antifungal agent, an endocrine disruptor, an EC 1.1.1.146 (11beta-hydroxysteroid dehydrogenase) inhibitor, an autophagy inducer and a calcium channel blocker. It is a polyphenol and an olefinic compound. |
|
Manufacturing Process |
50 parts by weight of p-hydroxypropiophenone are dissolved in 200 parts by weight of a 12.5% solution of caustic soda and shaken with 350 parts by weight of 3% sodium amalgam. The sodium salt of the pinacol thereby precipitating is reacted with glacial acetic acid, whereby the free pinacol is obtained (MP 205°C to 210°C, after purification 215°C to 217°C). The yield amounts to 95% of the theoretical. The pinacol is suspended in ether and gaseous hydrogen chloride introduced, whereby water separates and the pinacolin formed is dissolved in the ether, from which it is obtained by evaporation as a viscous oil (diacetateof MP 91°C). The yield is quantitative.40 parts by weight of pinacolin are dissolved in ethyl alcohol and gradually treated with 80 parts by weight of sodium under reflux. The solution is decomposed with water and the pinacolin alcohol formed extracted from the neutralized solution with ether. The pinacolin alcohol is a viscous oil which is characterized by a dibenzoate of MP 172°C. The yield is 95% of the theoretical.A solution of 30 parts by weight of pinacolin alcohol in ether is saturated with hydrogen chloride at room temperature and the ether solution then agitated with bicarbonate. After concentration by evaporation it leaves behind the crude diethylstilbestrol [α,β-(p,p'-dihydroxydiphenyl)-α,β-diethylethylene] which, when recrystallized from benzene, melts at 170°C to 171°C. The yield amounts to 75% of the calculated. The total yield of diethylstilbestrol, calculated on p-hydroxypropiophenone, is 68% of the theoretical. |
|
Brand name |
Stilbestrol (Tablicaps); Stilbetin (Bristol-Myers Squibb);Distilbene;Oestro-gynedron;Stilphostrol. |
|
Therapeutic Function |
Estrogen |
|
World Health Organization (WHO) |
Diethylstilbestrol, a synthetic estrogen which is a stilbene derivative, was introduced into obstetric practice in the late 1940s and subsequently widely used for the treatment of threatened abortion. This use was later shown to be associated with an increased risk of vaginal cancer in the offspring which resulted in restrictive regulatory action in several countries. Diethylstilbestrol and other stilbenes remain available in many countries, however, for the treatment of certain hormone-dependent neoplasms including carcinoma of the prostate and postmenopausal breast cancer. (Reference: (WHODI) WHO Drug Information, 77.1, 16, 1977) |
|
General Description |
Diethylstilbestrol is an odorless tasteless white crystalline powder. (NTP, 1992) |
|
Air & Water Reactions |
Insoluble in water. |
|
Reactivity Profile |
Diethylstilbestrol is incompatible with strong oxidizing agents, strong bases, acid chlorides and acid anhydrides . |
|
Fire Hazard |
Flash point data for Diethylstilbestrol are not available; however, Diethylstilbestrol is probably combustible. |
|
Biochem/physiol Actions |
Diethylstilbestrol is a synthetic estrogen with carcinogenic properties. Causes renal clear-cell carcinoma in Syrian hamster. In humans it causes increased risk of breast cancer, clear cell adenocarcinoma (CCA) of the vagina and cervix, and reproductive anomalies. Used in the treatment of prostate cancer to block the production of testosterone. |
|
Side effects |
These include sodium retention and oedema,nausea,gynaecomastia and impotence in men, and venous and arterial thrombosis. It can cause bone pain and hypercalcaemia when used for breast cancer. |
|
Safety Profile |
Confirmed carcinogen producing skin, liver, and lung tumors in exposed humans as well as uterine and other reproductive system tumors in the female offspring of exposed women. Experimental carcinogenic, neoplas tigenic, tumorigenic, and teratogenic data. A transplacental carcinogen. A human teratogen by many routes. Poison by intraperitoneal and subcutaneous routes. It causes glandular system effects by sktn contact. Human reproductive effects by ingestion: abnormalspermatogenesis; changes in testes, epididymis, and sperm duct; menstrual cycle changes or disorders; changes in female ferulity; unspecified maternal effects; developmental abnormalities of the fetal urogenital system; germ cell effects in offspring; and delayed effects in newborn. Implicated in male impotence and enlargement of male breasts. Other experimental reproductive effects. Mutation data reported. When heated to decomposition it emits acrid smoke and fumes. See also ETHINYL ESTRADIOL. |
|
Carcinogenicity |
Diethylstilbestrol is known to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in humans. |
|
Environmental Fate |
Diethylstilbestrol is a known teratogen and carcinogen. Experimental studies using transgenic estrogen receptor knockout animals suggest that binding and activation of the estrogen receptor is required to elicit diethylstilbestrol toxicity. Hence, diethylstilbestrol lesions primarily appear in tissues enriched with estrogen receptors. Diethylstilbestrol binds to the estrogen receptor with a very high affinity and forms a complex with the target tissue. The complex then internalizes in to the cell and translocates to the nucleus. Once in the nucleus, diethylstilbestrol may directly bind with the cellular DNA and cause mutations and unscheduled DNA synthesis. Diethylstilbestrol is also known to induce aneuploidy. |
|
Toxicity evaluation |
Diethylstilbestrol’s production and use in biochemical research, medicine, and veterinary medicine may result in its release to the environment through various waste streams. It may also be released to the environment during transport, storage, or disposal. If released to soil, diethylstilbestrol is predicted to strongly adsorb to the soil. Volatilization from the dry or wet soil surface would probably be unlikely. The extent of biodegradation in soil is not known, although diethylstilbestrol has been shown to be resistant to degradation in activated sludge. If released to water, diethylstilbestrol may bioconcentrate in aquatic organisms and strongly adsorb to suspended solids and sediments. Diethylstilbestrol is expected to be essentially nonvolatile on water surfaces. Diethylstilbestrol would not be susceptible to hydrolysis. The extent of biodegradation in natural waters is not certain, although diethylstilbestrol has been shown to be resistant to degradation in activated sludge. If released to the atmosphere, diethylstilbestrol vapors should rapidly oxidize, primarily by reaction with ozone. It is expected to exist solely in the particulate phase in an ambient atmosphere. Particulatephase diethylstilbestrol may be removed from the air by wet and dry deposition. |
|
Clinical claims and research |
At first glance, it might be surprising that synthetic nonsteroidal molecules such as diethylstilbestrol (DES) could have the same activity as estradiol or other estrogens. DES can be viewed, however, as a form of estradiol with rings B and C open and a six-carbon ring D. The activity of DES analogs was explained in 1946. It was proposed that the distance between the two DES phenol OH groups was the same as the 3-OH to 17-OH distance of estradiol; therefore, they could both fit the same receptor. Medicinal chemists have shown the OH-to-OH distance to be actually 12.1 ? in DES and 10.9 ? in estradiol. In aqueous solution, however, estradiol has two water molecules that are hydrogen bonded to the 17-OH. If one of the two water molecules is included in the distance measurement, there is a perfect fit with the two OH groups of DES. This suggests that water may have an important role for estradiol in its receptor site.It is now generally accepted that the estrogens must have a phenolic moiety for binding, but some investigators propose that the receptor may be flexible enough to accommodate varying distances between the two key hydroxyls. This point about estrogens needing a phenolic ring for high-affinity binding to the ER is critical. Steroids with a phenolic A ring and related phenolic compounds lack high-affinity binding to the other steroid hormone receptors. |
InChI:InChI=1/C18H20O2/c1-3-17(13-5-9-15(19)10-6-13)18(4-2)14-7-11-16(20)12-8-14/h5-12,19-20H,3-4H2,1-2H3/b18-17-
Due to its function as a regulator of dr...
We described the synthesis of flavone-es...
A novel pharmaceutical composition for t...
Monoclonal antibodies that bind specific...
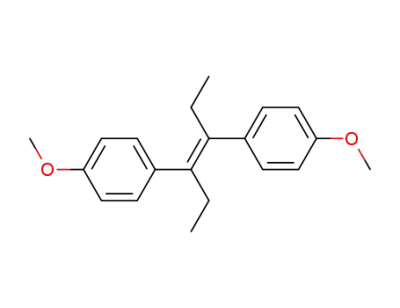
Diethylstilbestrol dimethyl ether

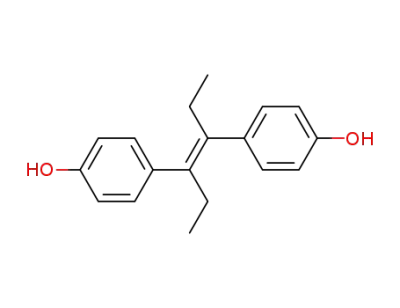
diethylstilbestrol
| Conditions | Yield |
|---|---|
|
Diethylstilbestrol dimethyl ether;
With
boron tribromide;
In
dichloromethane;
at -78 - 20 ℃;
for 2h;
Inert atmosphere;
With
water;
In
dichloromethane;
at 0 ℃;
for 0.5h;
|
63% |
|
With
boron tribromide-dimethyl sulfide complex;
In
1,2-dichloro-ethane;
at 83.5 ℃;
for 576h;
|
50.2% |
|
With
potassium hydroxide; Dipropylene glycol;
at 215 - 235 ℃;
|
|
|
With
potassium hydroxide;
at 200 ℃;
|
|
|
With
potassium hydroxide;
at 200 - 210 ℃;
|
|
|
With
potassium hydroxide; ethylene glycol;
at 180 - 215 ℃;
|
|
|
With
potassium hydroxide; diethylene glycol;
at 215 - 235 ℃;
|
|
|
With
potassium hydroxide;
at 200 - 210 ℃;
|
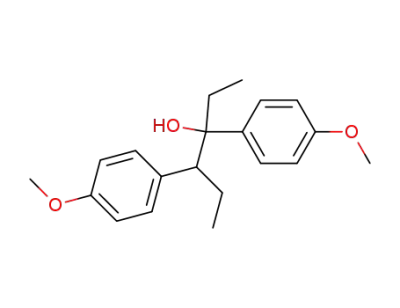
3,4-bis-(4-methoxy-phenyl)-hexan-3-ol


diethylstilbestrol
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: KHSO4 / 195 - 200 °C
2: ethanolic KOH / 200 - 210 °C
With
potassium hydroxide; potassium hydrogensulfate;
|
|
|
Multi-step reaction with 2 steps
1: PBr3 / 100 °C
2: 220 °C
With
phosphorus tribromide;
|
|
|
Multi-step reaction with 2 steps
1: KHSO4 / 210 °C
2: KOH; O-ethyl-diethylene glycol
With
potassium hydroxide; potassium hydrogensulfate; ethoxyethoxyethanol;
|
|
|
Multi-step reaction with 2 steps
1: PBr3 / 100 °C
2: 180 - 190 °C
With
phosphorus tribromide;
|
|
|
Multi-step reaction with 2 steps
1: KHSO4 / 210 °C
2: O-butyl-diethylene glycol
With
potassium hydrogensulfate; Diethylene glycol monobutyl ether;
|
|
|
Multi-step reaction with 2 steps
1: KHSO4 / 210 °C
2: O-ethyl-diethylene glycol
With
potassium hydrogensulfate; ethoxyethoxyethanol;
|
|
|
Multi-step reaction with 2 steps
1: PBr3 / 100 °C
2: 170 - 175 °C
With
phosphorus tribromide;
|
|
|
Multi-step reaction with 2 steps
1: PBr3; chloroform / 20 °C
2: 170 - 175 °C
With
chloroform; phosphorus tribromide;
|
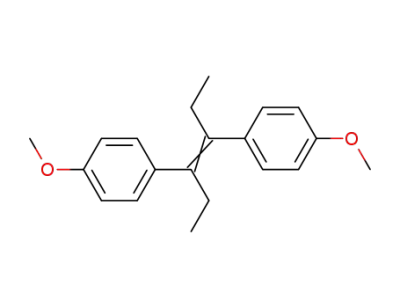
diethylstilbestrol dimethyl ether
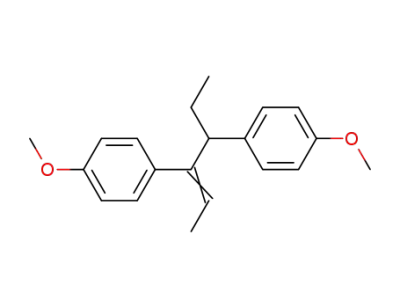
C20H24O2
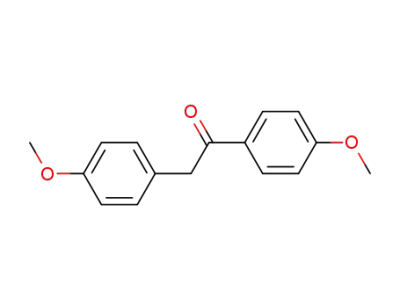
1,4-di(4-methoxyphenyl)ethanone
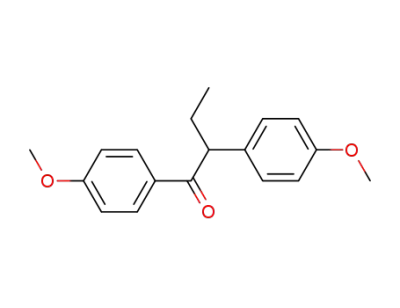
1,2-bis(4-methoxyphenyl)butanone
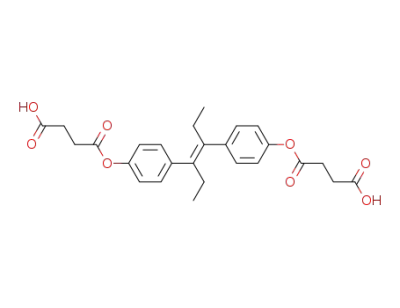
3,3'-<α,β-Diethyl-4,4'-stilbendiylbis-(oxycarbonyl)>-dipropionsaeure
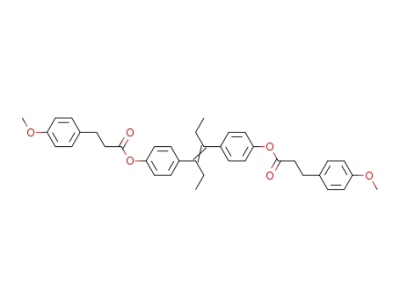
Diaethylstilboestrol-bis-β-(p-methoxyphenyl)-propionat
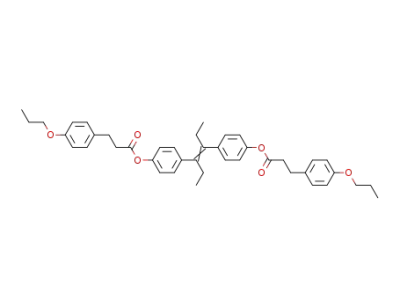
Diaethylstilboestrol-bis-β-(p-propoxyphenyl)-propionat
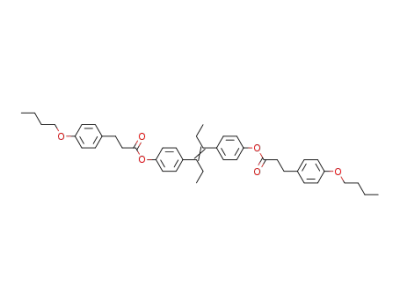
Diaethylstilboestrol-bis-β-(p-butoxyphenyl)-propionat